Isomers functions deal with various types of chemical isomerism such as:
- tautomerism
- stereoisomerism (tetrahedral, double bond)
Example 1
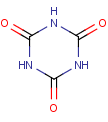
We examine the following molecule in terms of tautomerism:
This molecule is a triple lactam and can undergo lactam-lactim tautomerism. The possible tautomeric forms can be generated by CT:
allTautomers()
The following compounds are enumerated by this function as tautomer forms of the original molecule:
Let's see which one is the dominant tautomer form on pH=2.
dominantTautomer(0, "2.0")
THIS EXPRESSION DOES NOT WORK WITH CT!
Example 2
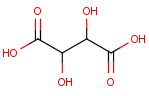
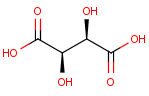
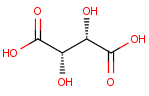
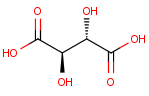
We calculate the number of stereoisomers for tartaric acid:
tetrahedralStereoisomerCount()
This function returns 3 corresponding to the following structures:
Alternatively, stereoisomerCount() function can also be used here.
Example 3
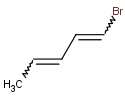
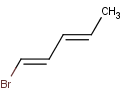
Let's study double bond stereoisomerism of this compound:
doubleBondStereoisomer(0)
This expression generates the first double bond stereoisomer of the input molecule as we use 0-based indexing in Chemical Terms: